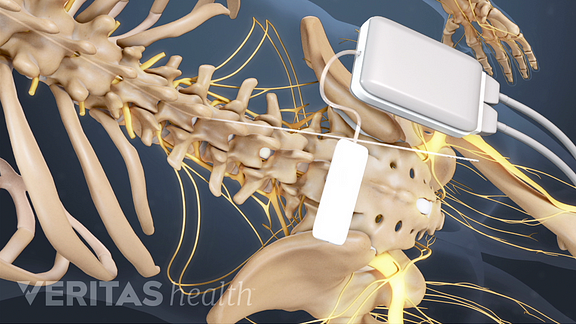
Last December CVS Health (NYSE: CVS) revealed its plans to buy Aetna (NYSE: AET) in a $69 billion merger that’s still pending. These decisions build on the momentum of broader national coverage decisions by private insurers and support the FDA’s goals to find new ways to combat the opioid epidemic with alternatives such as Abbott’s neurostimulation therapies.” “While Medicare already covers our DRG system, it’s encouraging to see private payers like Aetna review the clinical data and outcomes, then choose to provide access to DRG stimulation for their members. One of the most challenging stories we hear is when patients undergo a therapy trial, find pain relief and then learn their insurance won’t cover their therapy,” neuromodulation VP Keith Boettiger said in prepared remarks. “Since its launch in the U.S., Abbott’s DRG stimulation has had a profound impact for people living with complex pain conditions. The design of the IonicRF Generator enables healthcare teams to quickly become proficient in its use. It acquired the neurostim assets when it paid $25 billion for St. Aetna already covers treatment with Abbott’s spinal cord stimulation devices, the company said. The national coverage decision opens DRG treatment to some 22 million Aetna members, Abbott said. Federal law restricts this device to sale by or on the order of a physician.Abbott (NYSE: ABT) said today that it won a reimbursement nod from Aetna (NYSE: AET) for its Proclaim dorsal root ganglion neurostimulation therapy for treating chronic pain. For complete indications for use, contraindications, warnings, precautions, and side effects, call 866.360.4747 or visit .Ĭaution: U.S. Your doctor may be able to provide additional information on the Boston Scientific Spinal Cord Stimulator systems. subperception therapy) it is less likely that sudden stimulation changes resulting in distraction could occur while having stimulation on when operating moving vehicles, machinery, and equipment. For therapy that does not generate paresthesia (i.e. Food and Drug Administration (FDA) has approved its new Proclaim Plus spinal cord stimulation (SCS) system featuring FlexBurst360 therapy. Stimulation must be turned off first in such cases. 23, 2022 /PRNewswire/ - Abbott (NYSE: ABT) today announced that the U.S. Patients using therapy that generates paresthesia should not operate motorized vehicles such as automobiles or potentially dangerous machinery and equipment with the stimulation on. Advise your physician that you have a Spinal Cord Stimulator before going through with other implantable device therapies so that medical decisions can be made and appropriate safety measures taken. All neuromodulation patients should review this compatibility guide from Abbott prior to installing the NeuroSphere Patient Controller App on. 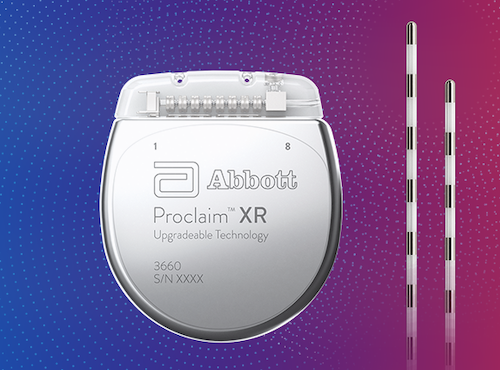
Supported devices by application and recommended iOS version are listed in the table below. The Spinal Cord Stimulator system may interfere with the operation of implanted sensing stimulators such as pacemakers or implanted cardiac defibrillators. Abbott has tested mobile devices and operating systems for compatibility with the patient controller app. In a multicenter study of BurstDR spinal cord stimulation, 100 of participants on a low-energy programming settings felt full pain relief on less than six hours of battery use per day, and 43 of participants achieved pain relief on less than 2 hours of battery use per day. 
The system should not be charged while sleeping. Strong electromagnetic fields, such as power generators or theft detection systems, can potentially turn the stimulator off, or cause uncomfortable jolting stimulation. As a Spinal Cord Stimulation patient, you should not have diathermy as either a treatment for a medical condition or as part of a surgical procedure. Exposure to MRI may result in dislodgement of the stimulator or leads, heating of the stimulator, severe damage to the stimulator electronics and an uncomfortable or jolting sensation. Patients implanted with Boston Scientific Spinal Cord Stimulator Systems without ImageReady™ MRI Technology should not be exposed to Magnetic Resonance Imaging (MRI).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
